Many forms of teeth whitening treatments have been used by dentists to remove years of stains from patients’ smiles. Although it’s considered safe, whitening treatments can leave some patients with a temporary sensation of tooth sensitivity. So what is the reasoning for this? Most often it is connected to peroxide whiteners and dentinal tubules.
Reasons for Increased Sensitivity
There are multiple causes of tooth sensitivity, and the level of pain can vary across patients. Theories speculate the strong role of whitening agents on dentinal tubules and the mechanisms that cause hypersensitivity. Studies show that acidic diets and low salivary pH levels demineralize peritubular dentin, often resulting in doubling the diameter of dentinal tubular orifices, as well as flaring the orifices similar to a trombone. Obviously, larger diameter and flared tubular orifices are not as likely to be closed with smear plugs, and once smear plugs are formed, they are dislodged far more easily, which increases sensitivity.
All peroxide-based whitening gels often lead to increased sensitivity through the dislodging of smear plugs. When smear plugs are removed, whitening gels come into direct contact with dentinal tubular fluids. The difference in osmolality between tubular fluid and whitening gel results in an osmotic gradient pulling dentinal tubular fluid outward, resulting in sensitivity.
Due to their hypertonicity, whitening gels range anywhere from 17 to 190 times higher in osmolality than natural dentinal tubular fluid. The greater the osmolality a whitening gel contains, the stronger the osmotic gradient will be between the gel and the tubular fluid, the more “pull” on the tubular fluid, and the more discomfort is experienced by the patient.
Chemical Stabilizers
Acidifiers (phosphoric acid) and anhydrous gels are common chemical stabilizers used by whitening gel manufacturers because they reduce the need for constant refrigeration. This greater chemical stability caused by the addition of these chemical stabilizers causes two significant concerns:
- Acid and anhydrous gel ingredients increase the osmolality of whitening gels up to 11 times higher than 100% aqueous and neutral pH gels. This causes up to 11 times more “pull” on dentinal tubular fluid, and significantly greater teeth sensitivity.
- By causing the whitening gels to be more chemically stable, they are not as unstable as we would like when placed in the mouth. The more unstable the whitening gels are in the mouth, the more thoroughly and effectively they release the whitening byproducts of any type of peroxide.
Science has shown that constant refrigeration is by far the best way to retain maximum potency in peroxide gels without the addition of chemical stabilizers that decrease whitening effectiveness and increase the risk of sensitivity.

Addition of Desensitizers to Whitening Gels
Some whitening manufacturers use fluoride, potassium nitrate and/or ACP in their whitening gels. However, studies show that, when mixed into the whitening gel formulation, none of these substances have proven to decrease sensitivity. Peroxide gels must have aggressive chemistry and a fast oxygenation process to be effective. This means that any orderly, passive formation of dentinal tubular plugs by fluoride or ACP is entirely interrupted by the aggressive oxygenation chemical reaction of the peroxide gels.
Potassium nitrate, on the other hand, does not plug dentinal tubules. It must migrate through the dentinal tubules and into the pulp of the teeth to have any positive benefit. The osmotic gradient produced by all whitening gels causes dentinal tubular fluid to flow outward, away from the pulp. Therefore, potassium nitrate would have to somehow, “swim upstream against the current” to reach the pulp during the whitening process, which is scientifically unlikely.
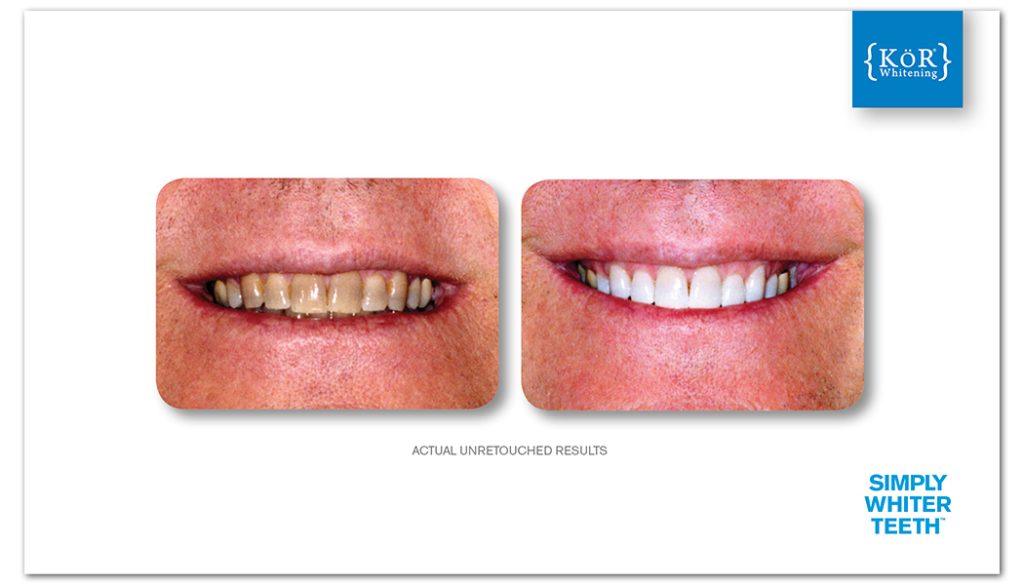
The KöR® Whitening System
The scientific literature is generally in agreement that the best way of handling whitening gels is constant refrigeration, from the instant of manufacture until received cold by the dental practice, instead of use of chemical stabilizers.
This provides not only whitening gels that are at virtually 100% of the original potency but also results in exceptionally unstable gels when placed in the mouth, leading to extraordinary whitening effectiveness. With no need for chemical stabilizers (because of constant refrigeration), the osmolality is as low as 1/11th compared to other whitening gels, resulting in significantly less pull on dentinal tubular fluid, and less likelihood of sensitivity.
Additionally, the scientific literature is also generally in agreement that desensitizers should be used before and/or after whitening gel applications, however, should not be mixed within the whitening gel formulation itself.
It is important to understand the science behind teeth whitening in order to produce a positive treatment experience for patients. That’s why KöR® Whitening’s solutions are based in scientific research, to create reliable and results-oriented whitening solutions. To learn more about the KöR® Whitening system and how to provide our products to your patients, contact us today.